On October 31, 2023, the alternative medicine community woke up to unprecedented positive data demonstrating the safety and effectiveness of psilocybins in the treatment of depression.
Cybin, a biotechnology company working on next-generation psychedelic treatments, announced remarkable results from its Phase 2 clinical trial using a proprietary version of deuterated psilocybin for the treatment of depression. CYB003 is a next-generation psilocybin designed to provide a faster onset of action and to reduce the duration of effects, thereby reducing the financial barrier to accessing this medication in a medical setting.
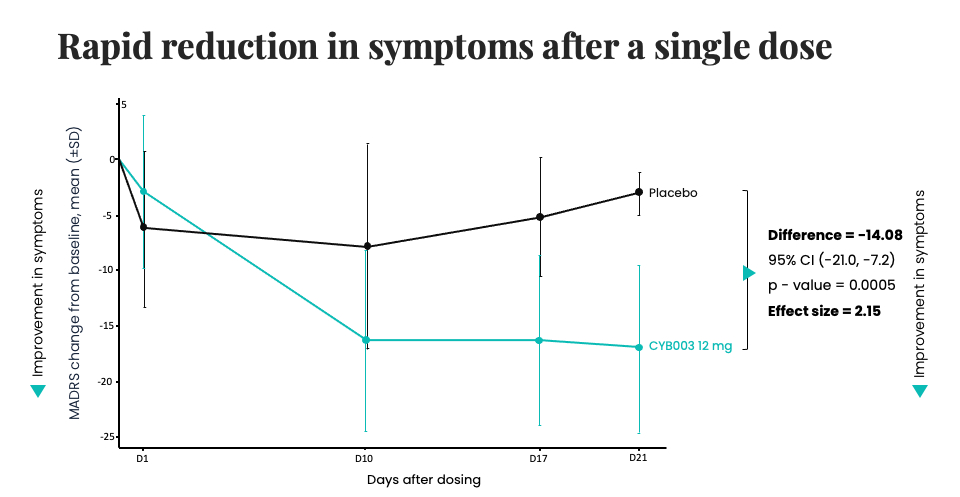
Interim trial data demonstrated rapid, robust, and clinically meaningful reduction in depression symptoms three weeks after a single dose of 12 mg of CYB003.
Specifically, three weeks after a single administration of the drug, 53.3% of patients experienced at least a 50% reduction in depressive symptoms (50% reduction in MADRS) and 20% of participants entered remission, which which means that they no longer met the clinical definition. of depression.
Results demonstrate a 14-point reduction in Montgomery-Asberg Depression Rating Scale (MADRS) score after administration of CYB003, compared to 1.82 points for traditional antidepressants compared to placebo. The marked difference in effectiveness is what makes the results truly unprecedented.
The speed of these results should not be overlooked. Typically, a significant reduction in depression symptoms takes more than three weeks, making CYB003’s onset of action faster than conventional treatments, which can take more than several weeks to simply show any effect.
Provided Cybin is able to replicate these results in its Phase 3 trials, we could see another depression solution on the market in the coming years.
The overwhelmingly positive interim results for the 12 mg dose of CYB003 are extremely encouraging for patients and providers. The demonstrated effectiveness at this dosage showed an unprecedented reduction in depressive symptoms compared to currently available treatments, said Doug Drysdale, CEO of Cybin. With these encouraging results in hand, we look forward to sharing the full baseline data later this quarter, as well as 12-week sustainability data in Q1 2024. Our planning continues as we prepare for a trial of phase 3 international and larger multi-site. in early 2024 to further evaluate the safety and effectiveness of CYB003 in people with MDD.
Cybin is not the only company making progress through clinical trials. Earlier this year, Compass Pathways began recruiting for the largest Phase 3 clinical trial investigating the safety, effectiveness and tolerability of psilocybin in the treatment of treatment-resistant depression (TRD), totaling 946 participants, and is expected to complete the study by 2025. If Compass delivers positive data, we could see FDA approval for psilocybin treatment in late 2025.
#Single #dose #nextgeneration #psilocybin #shows #significant #reduction #depression #longer #needing #treatment #Spotlight #psychedelics
Image Source : psychedelicspotlight.com